
Drug development, from initial discovery of a promising target to the final medication, is an expensive, lengthy, and incremental process. With trials that can take upwards of over 10 years to complete, reducing the time and cost of the R&D process as a whole, and of clinical trials in particular, is of utmost importance.
The more money companies must invest, the more risk there is of losing investment, and the longer this process drags on, the more likely the company will lose its competitive advantage. So what can be done to accelerate pharmaceutical drug discovery and clinical trials?
Combining Total Brain’s international neuroscientific database, comprised of more than 1.3M standardized datasets, along with our robust mental health monitoring and support platform, provides pharmaceutical companies a way to more easily monitor and track patient progress during clinical trials.
These datasets can be licensed in whole, or in part, to support your companies’ clinical studies and precision drug development efforts.

Total Brain’s clinically validated, digital assessment enables monitoring of cognitive and emotional functioning via 12 brain capacities, with integrated clinical screenings for 7 mental conditions like Addiction (CAGE-AID), Depression (PHQ-9), Anxiety (GAD7) and PTSD (PC-PTSD-5), among others.
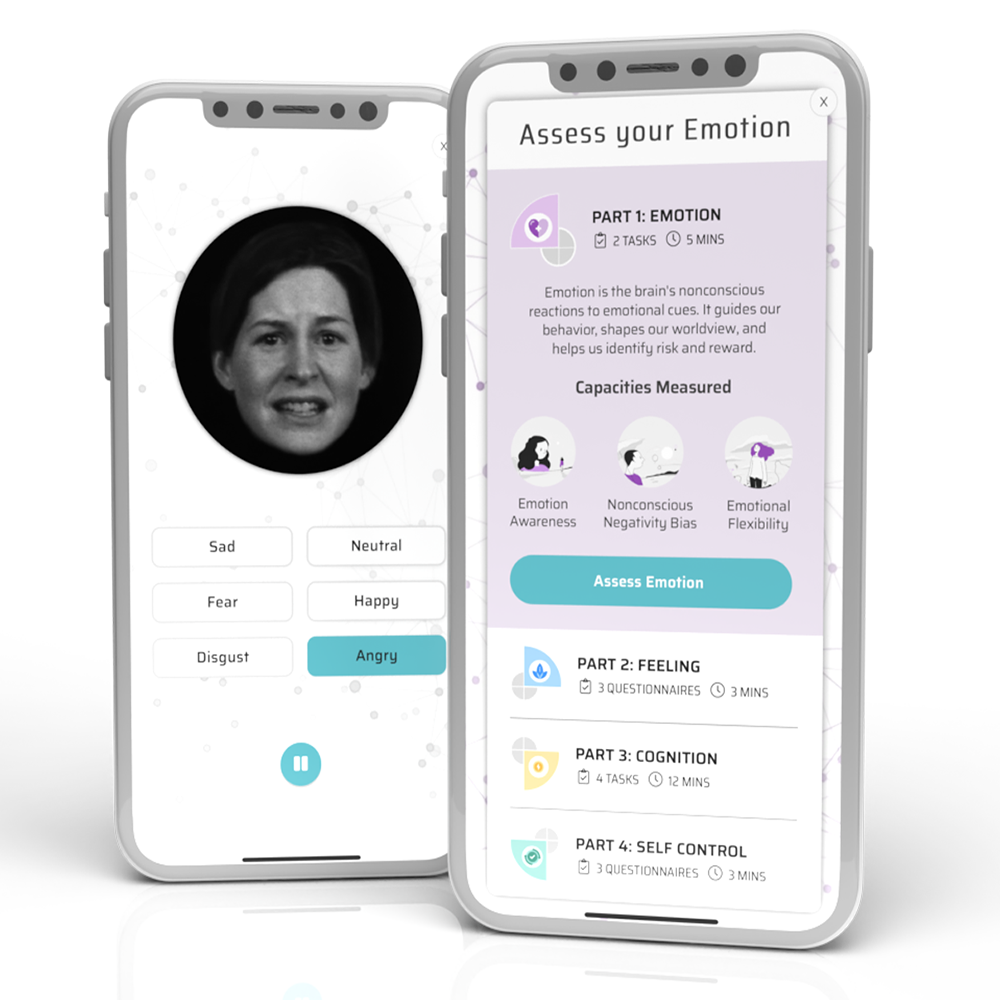
The assessment is designed to be repeated monthly, with results feeding a clinician dashboard to provide ongoing, objective patient data during clinical trials. Total Brain’s clinical support dashboard:
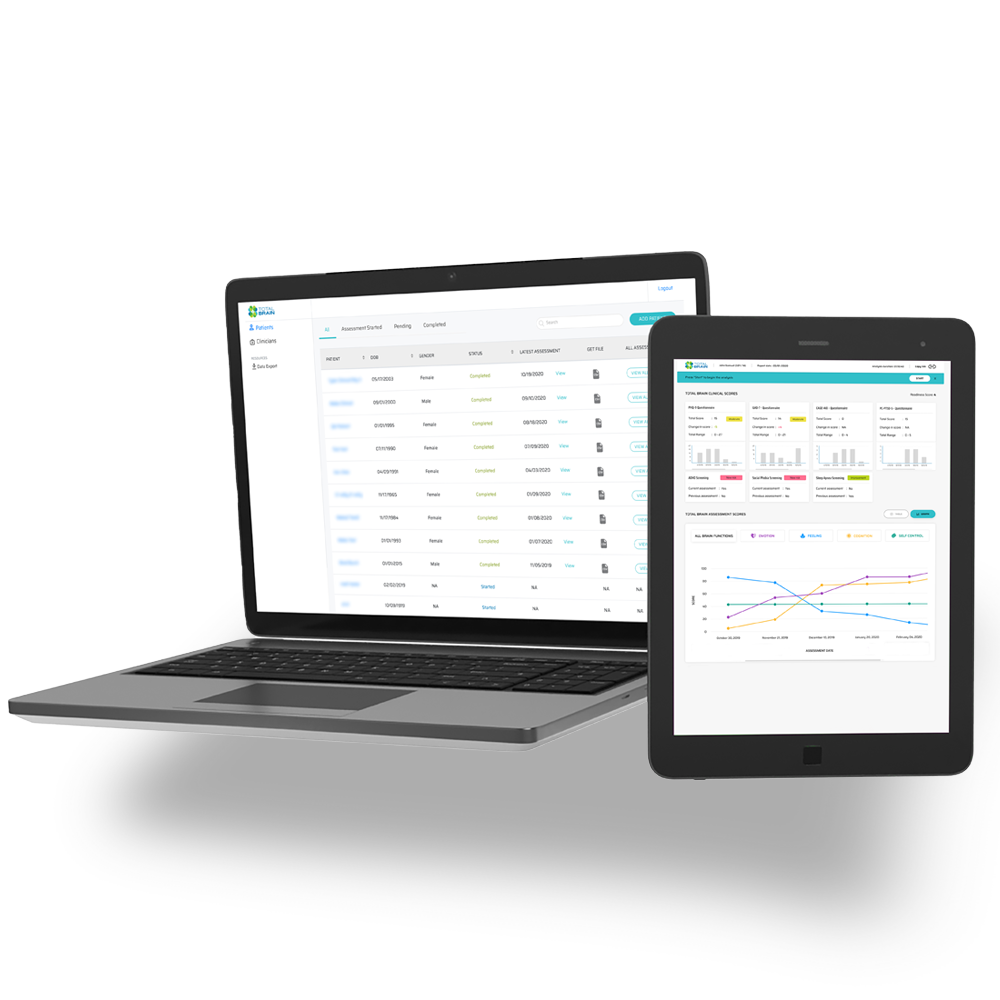
See how Total Brain can help you
Copyright © 2019 Total Brain. Terms and Conditions | Privacy | FAQ